2,4-Dichlorobenzoyl Chloride
Home / 2,4-Dichlorobenzoyl Chloride
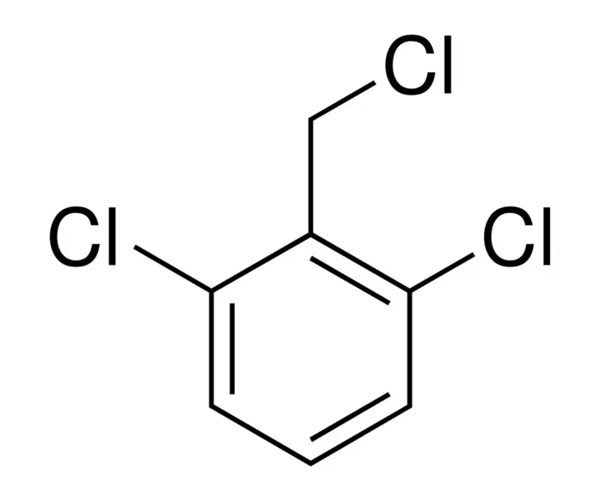
2,4-Dichlorobenzoyl Chloride Manufacturer Supplying High-Purity Aromatic Acid Chloride for Pharmaceutical and Specialty Chemical Production
Finding a reliable 2,4-Dichlorobenzoyl Chloride Manufacturer is essential for industries that need high-quality building blocks for complex chemistry. This compound is a clear to yellowish liquid that belongs to the aromatic acid chloride family. It serves as a vital starting point for creating various advanced molecules. Because it is highly reactive, it allows chemists to attach specific chemical groups to other structures easily, making it a favorite in large-scale industrial labs.
As a key Aromatic Acid Chloride Intermediate, this chemical plays a major role in the creation of modern medicines and specialized crop protection products. It acts like a chemical “bridge,” helping to link different molecules together to form final products that are stable and effective. Many Specialty Chemical Production plants rely on this specific chloride to ensure their end products meet the strict purity standards required for global markets.
Chemical Identification Table
Feature | Details |
Product Name | 2,4-Dichlorobenzoyl Chloride |
Chemical Formula | C7H3Cl3O |
CAS Number | 89-75-8 |
EC Number | 201-935-9 |
Molecular Weight | 209.46 g/mol |
UN Number | 3265 (Corrosive liquid, acidic, organic, n.o.s.) |
Appearance | Clear to light yellow liquid |
Chemical Type | Aromatic Acid Halide |
Physical and Chemical Properties Table
Property | Value / Description |
Boiling Point | Approximately 150°C at 20 mmHg |
Melting Point | Around 15°C to 18°C |
Density | 1.49 g/cm³ at 25°C |
Flash Point | Above 110°C |
Solubility Behavior | Reacts with water and alcohol; soluble in organic solvents |
Chemical Stability | Stable under recommended storage; keep dry |
Reactivity Profile | Highly reactive with moisture, bases, and oxidizing agents |
Why Choose Shital Chemical Industries
Choosing a trusted Chemical Intermediate Manufacturer ensures that your production line never stops. Here is why we stand out:
Process-Controlled Manufacturing
Carefully controlled chemical synthesis operations to ensure every drop meets high standards.
Assured Purity Consistency
Consistent product purity across manufacturing batches, so your results are always the same.
In-House Analytical Testing
Dedicated laboratory testing and analytical verification using modern equipment.
Reliable Bulk Production Capability
Strong infrastructure for bulk chemical production to handle large orders easily.
Hazardous Chemical Handling
Expertise handling reactive aromatic acid chloride compounds with maximum safety.
Reliable Global Supply
Reliable supply for both domestic and global buyers through a strong network.
Packaging and Export Capability
As a leading 2,4-Dichlorobenzoyl Chloride Supplier, we ensure that our logistics are as high-quality as our chemistry:
Industrial Drum Packaging
Industrial drum packaging specifically designed to hold hazardous liquids safely.
Moisture-Protected Sealing
Moisture-protected sealed containers to prevent the chemical from reacting before it reaches you.
Hazard labeling Standards
Hazard labeling that follows all international transport regulations for clear identification.
International Transport Classification
Compliance with global chemical shipping standards for smooth customs clearance
Export Documentation
Full export documentation, including a Certificate of Analysis (COA) and MSDS.
Global shipping Capability
Reliable worldwide logistics ensuring timely delivery to your facility.
Applications of Chloroacetyl Chloride
This 2,4-Dichlorobenzoyl Chloride Chemical is a foundation stone for many life-saving drugs. It is used by pharmaceutical manufacturing companies to create the core structure of various medicines, especially those targeting infections or inflammation. Because it reacts so predictably, it helps drug makers build complex molecules step-by-step without losing purity.
In the medical world, having a clean starting material is non-negotiable. Using this high-purity intermediate allows labs to produce active ingredients that are safe for human use. It is frequently found in the synthesis of specialized antibiotics and other therapeutic agents that require a chlorinated benzene ring as part of their structure.
In the world of farming, this compound is used to create powerful herbicides and fungicides. Agrochemical production plants use it to build molecules that protect crops from pests and diseases. By using this intermediate, manufacturers can create products that are more effective at lower doses, which is better for the environment and the farmer.
These chemicals are essential for maintaining global food supplies. The chlorine atoms on the ring provide specific biological activities that help stop weeds from growing or prevent mold from destroying harvests. It is a key ingredient for companies focused on high-tech crop protection.

General organic chemistry labs use this acid chloride to perform “acylation” reactions. This is a process where the benzoyl group is attached to another molecule. It is a very common tool used by researchers to explore new chemical structures or to create custom molecules for specific industrial tasks.
Because it is a liquid, it is easy to measure and pump in a factory setting. This makes it a preferred choice for companies that produce a wide variety of organic compounds on a daily basis. It serves as a versatile tool in the kit of any industrial chemist.

Specialty chemical laboratories often require this compound to make unique additives or stabilizers. These small-batch chemicals might be used in high-performance coatings or specific cleaning agents. The 2,4-dichloro structure provides a unique set of physical properties that other chlorides cannot match.
These specialty products are often sold to other industries that need very specific performance traits. By starting with a high-quality intermediate, these labs can guarantee that their final specialty product will perform exactly as their customers expect.

The polymer and material industries use aromatic acid chlorides to create high-strength plastics and resins. When reacted with other chemicals, it can help form long chains that result in materials that are resistant to heat and chemicals. This makes it useful for creating parts for cars or electronics that need to last a long time.
These resins are often used in tough environments where regular plastic would melt or break. By including a chlorinated aromatic ring in the polymer backbone, the resulting material becomes much more durable and rigid.

The color industry relies on this compound to create bright and lasting dyes. Fine chemical manufacturers use it to build the “chromophore,” which is the part of a molecule that gives it color. This is especially important for dyes used in textiles and high-end printing inks.
Because the chlorine atoms affect how the molecule absorbs light, they help create specific shades that stay vibrant even after washing or exposure to the sun. This makes it a valuable asset for companies producing industrial-grade colorants.

FAQs
What is 2,4-Dichlorobenzoyl Chloride used for?
It is primarily used as a building block to create medicines, farm chemicals like weed killers, and high-performance dyes or plastics.
Is this chemical hazardous?
Yes, it is a corrosive liquid that can cause burns to the skin and eyes. It also reacts strongly with water, so it must be handled with great care using safety gear.
How should 2,4-Dichlorobenzoyl Chloride be stored?
It must be stored in a cool, dry, and well-ventilated area. Keep the containers tightly sealed and away from moisture, heat, and open flames.
Which industries commonly use this compound?
The most common users are pharmaceutical companies, agrochemical plants, plastic manufacturers, and factories that make industrial dyes.
Can 2,4-Dichlorobenzoyl Chloride be supplied in bulk quantities?
Yes, as a large-scale manufacturer, we provide this chemical in industrial-sized drums and bulk containers for global export.
Why is this compound important in aromatic chemical synthesis?
Its high reactivity allows it to easily link with other molecules, making it an efficient “bridge” for building complex aromatic structures in a lab.

